|
Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Use the information below to generate a citation. A carbon-12 atom has 6 protons and 6 neutrons, so the mass of a proton or a neutron works out to be approximately 1 amu. One atomic mass unit (amu) is defined as 1/12 the mass of a Carbon-12 atom. Then you must include on every digital page view the following attribution: Atomic mass units (or atomic weight units) are then used to measure the mass of atoms. If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the For example, Copper (Cu) has two stable isotopes: Along with Johannes Jensen and Eugene Wigner, Maria Goeppert Mayer received the Nobel Prize for this work, becoming the second woman to win the award.īecause of the existence of stable isotopes, we must take special care when quoting the mass of an element. 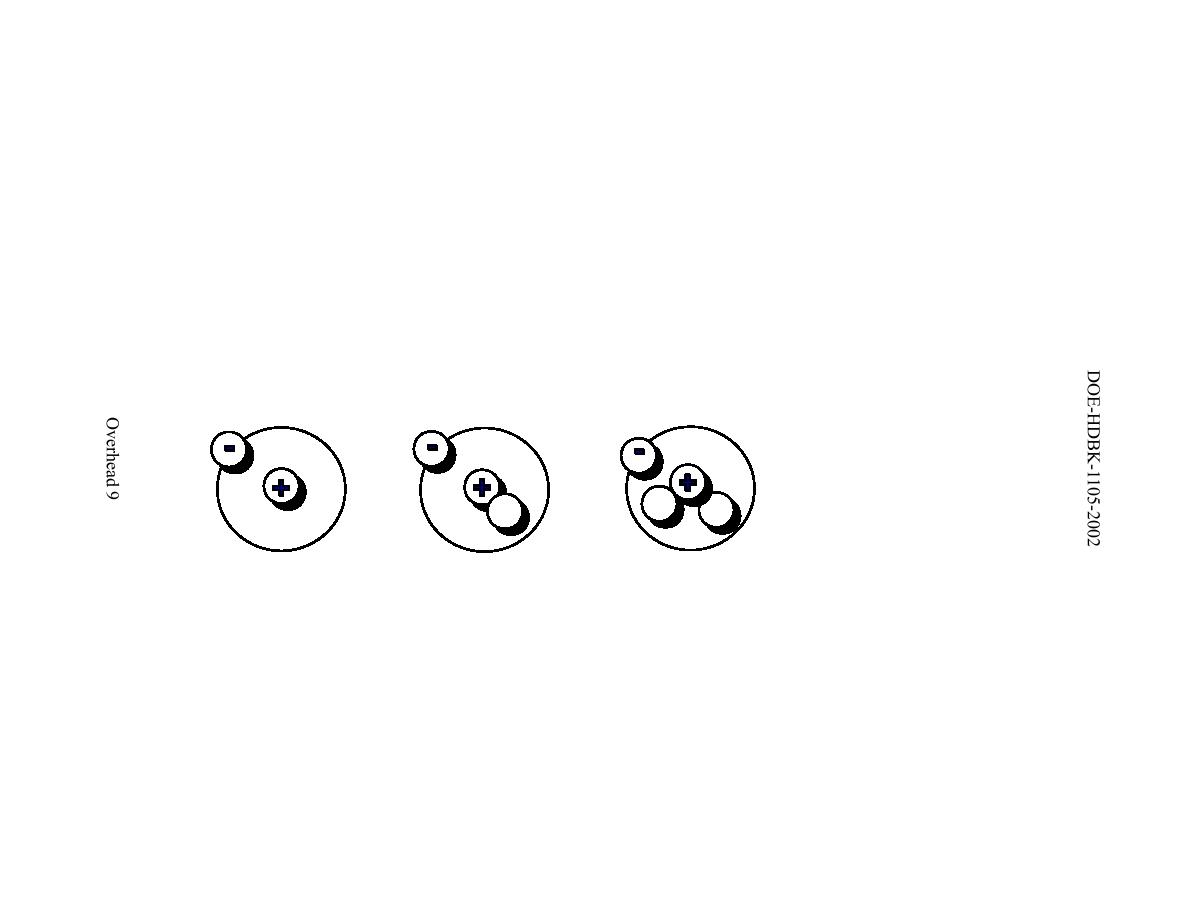
The theory has been very successful in explaining nuclear energy levels, nuclear decay, and the greater stability of nuclei with closed shells. Goeppert Mayer and other researchers recognized that "closed" nuclear shells are more stable than others. German-born physicist Maria Goeppert-Mayer identified these characteristics based on certain quantities of nucleons, leading to her development of nuclear shell theory. The arrows represent attractive forces exerted by a neutron (in red) on its nearest neighbors.ĭetailed examination reveals greater stability and more attractive nuclear forces when when neutrons and protons are in pairs. 
(b) The strong nuclear force acts between neighboring nucleons. The arrows represent outward forces on protons (in blue) at the nuclear surface by a proton (also in blue) at the center. They are equal to 16, 16, and 18, respectively. Find the numbers of protons, neutrons, and electrons. Let's assume that it is the sulfide anion. For heavy nuclei ( Z > 15 ), ( Z > 15 ), excess neutrons are necessary to keep the electrostatic interactions from breaking the nucleus apart, as shown in Figure 10.3.įigure 10.5 (a) The electrostatic force is repulsive and has long range. Alternatively, you can also calculate the atomic number, atomic mass, and charge. Nuclear stability occurs when the attractive forces between nucleons compensate for the repulsive, long-range electrostatic forces between all protons in the nucleus. We discuss strong nuclear force in more detail in the next chapter when we cover particle physics. This force is responsible for some nuclear decays, but as the name implies, it does not play a role in stabilizing the nucleus against the strong Coulomb repulsion it experiences. You may also have heard of a “weak” nuclear force. U-238 has 92 protons and 146 neutrons, while U-235 has 92 protons and 143 neutrons. Carbon-14, for example, contains 6 protons + 8 neutrons to make the atomic mass number of 14. 
Two types of forces exist: (1) the long-range electrostatic (Coulomb) force that makes the positively charged protons repel one another and (2) the short-range strong nuclear force that makes all nucleons in the nucleus attract one another. In all cases, the number of protons + the number of neutrons atomic mass number of an atom. Tritium is a radioactive species having a half-life of 12. Its nucleus, consisting of one proton and two neutrons, has triple the mass of the nucleus of ordinary hydrogen. Why do neutrons outnumber protons in heavier nuclei ( Figure 10.5)? The answer to this question requires an understanding of forces inside the nucleus. Tritium, (T, or 3H), the isotope of hydrogen with atomic weight of approximately 3. \): Formalism used for identifying specific nuclide (any particular kind of nucleus) The atomic symbol, X, is the abbreviation used to represent an atom in chemical formulas.Table 10.1 Common Isotopes *No entry if less than 0.001 (trace amount).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
